Hello,
This is the 3rd in a series of 3 posts related to cancer genetics. This post focuses on the genetics and risks associated with BRCA variants.
For those attending ACMG or SIMD this week, I hope you have a great time. There will be members of the StudyRare team (residents and fellows from Colorado) who will be attending both conferences. We will have some StudyRare stickers and business cards to share!
Please feel free to email with any comments or questions about this post (daniel@studyrare.com).
Also, please share this post with anyone who might find it useful!
-Daniel
Questions
Question 36
A 31-year-old woman presents to her primary care physician with concerns about her risk for developing breast and ovarian cancer. She has a strong family history of both diseases, with her mother being diagnosed with breast cancer at age 43 and her grandmother being diagnosed with ovarian cancer at age 57. Genetic testing reveals that the patient is positive for a pathogenic variant in the BRCA2 gene. In addition to her risk for developing breast and ovarian cancer, she is also at increased risk for developing which of the following types of cancer?
Question 37
The patient in question 36 is planning her first pregnancy and desires to know her reproductive risks related to the BRCA2 variant. Her husband’s family history is notable for breast cancer in his father. Her husband undergoes genetic testing, which is also positive for a BRCA2 variant. A child who inherits both BRCA2 variants would be at risk for presenting with which of the following symptoms?
Question 38
The BRCA2 gene encodes a protein involved in which of the following cellular processes?
Explanation
BRCA2 variants are a major risk factor for hereditary breast and ovarian cancer (HBOC). In addition to breast and ovarian cancer, patients with BRCA2 variants have an increased risk for other cancer types such as pancreatic carcinoma, prostate cancer, and melanoma (Question 36). BRCA1 variants are also associated with breast, ovarian, pancreatic, and prostate cancer but do not appear to increase the risk of melanoma. Though additional (and weaker) associations are being identified between BRCA genes and other cancers through ongoing research, these are the most important associations to know for counseling patients and for board exams.
Biallelic BRCA1/2 variants are associated with Fanconi anemia, a clinically-distinct recessive disorder
When both parents are carriers of a BRCA1 or BRCA2 variant, their offspring may develop Fanconi anemia (FA) if they inherit biallelic variants in either BRCA1 or BRCA2. Of note, FA can be also caused by biallelic variants in other genes involved in DNA repair (most commonly FANCA). FA is an autosomal recessive disorder characterized by short stature, bone marrow failure leading to cytopenias, and an increased risk of childhood-onset cancers (Question 37). Patients also have congenital anomalies including hypoplastic thumb and forearm along with kidney, heart, eye, and genital anomalies. The risk for developing a variety of cancers early in life is also significantly increased in FA. For example, in patients with BRCA2-related Fanconi anemia, the cumulative probability of any malignancy (most commonly leukemia, medulloblastoma, or Wilms tumor) was 97% by age 6 years. There is currently no cure for FA, and management options include bone marrow transplant, blood transfusions, and cancer surveillance.
The concept of recessive disorders in genes that predispose to cancer (and that are also involved in DNA repair) is an important concept to be aware of. Below are some more examples of genes involved in DNA repair that are associated with both an autosomal dominant cancer predisposition and a clinically-distinct and more severe autosomal recessive disorder:
Monoallelic variants in ATM are associated with increased risks for breast, ovarian, and pancreatic cancers, while biallelic variants in ATM are associated with the autosomal recessive disorder ataxia-telangiectasia (discussed below).
Monoallelic variants in genes involved in DNA mismatch repair (MLH1, MSH2, MSH6, or PMS2) predispose to colorectal and endometrial cancer (among others), while biallelic variants in these same genes cause constitutional mismatch repair deficiency (CMMRD), a syndrome that presents with multiple cafe-au-lait macules, Lisch nodules, and freckling (similar to NF1) along with multiple cancers in childhood.
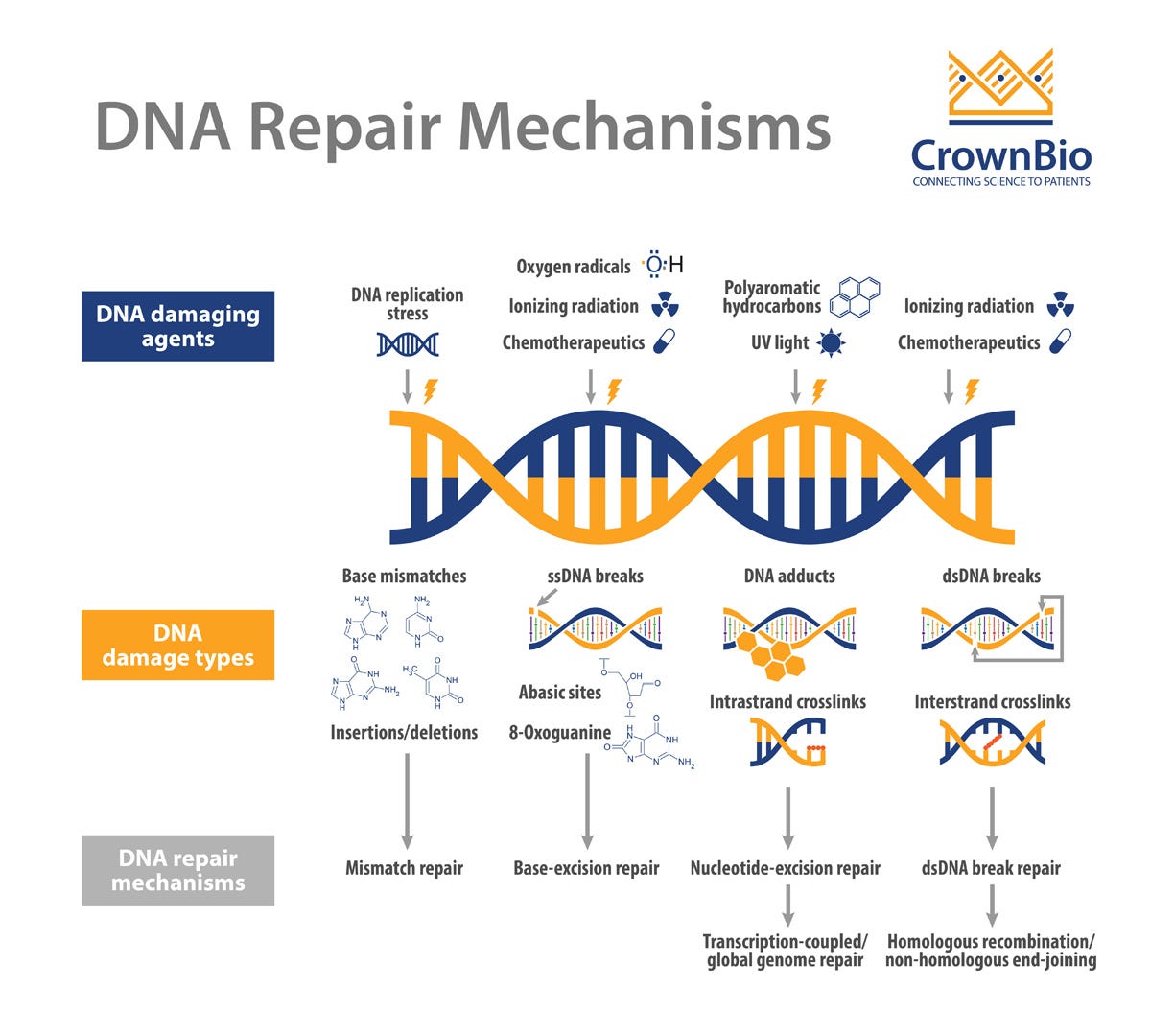
DNA repair mechanisms
Broadly, there are 4 main DNA repair mechanisms: double-strand DNA break repair, DNA mismatch repair, base excision repair, and nucleotide excision repair (for more info, see Khan Academy, this Twitter thread, and the figure below). The BRCA1/2 genes encode for proteins involved in double-strand DNA break repair (dsDBR) (Question 38). dsDBR is carried out via 2 main mechanisms: homologous recombination (HR) and non-homologous end joining (NHEJ). BRCA1 & BRCA2 are involved in HR, which uses the homologous DNA strand as a template to repair the double-stranded DNA breaks. In contrast, NHEJ pastes broken DNA strands together without using a template. Generally, DNA repair mechanisms are critical for maintaining genomic stability and preventing the accumulation of harmful mutations that can lead to cancer. In patients with pathogenic variants in BRCA1 or BRCA2, mutations accumulate in cells and the risk of cancer increases because dsDBR is impaired.
💡 The 4 main DNA repair mechanisms “Make DNA Better:” Mismatch repair, Double-stranded DNA break repair, Nucleotide excision repair, Base excision repair
Incorrect answers (Question 37)
Hemihypertrophy and omphalocele are associated with Beckwith-Weidemann syndrome (BWS). Patients with BWS may also have macrosomia, a large tongue, and ear lobe creases. Pediatric patients have an increased risk of embryonal tumors (e.g. Wilms tumor, hepatoblastoma, neuroblastoma, rhabdomyosarcoma) and adrenocortical carcinoma. Of note, the risk for tumors in BWS is greatest in early childhood and decreases thereafter, whereas the risk for BRCA1/2-related cancer is low in childhood and increases in adulthood.
Immunodeficiency (presenting as recurrent sinopulmonary infections), progressive ataxia, and telangiectasias (small dilated blood vessels) are features of ataxia-telangiectasia (AT), an autosomal recessive disorder caused by variants in the gene ATM, which encodes for a protein involved in NHEJ. The most common cancers in patients with AT are lymphoma and leukemia, though patients are also at risk for developing a variety of other cancers, including breast, ovarian, and thyroid cancer, among others. Patients who are heterozygous carriers of ATM variants also have an increased risk of breast, ovarian, pancreatic, and other cancers.
Macrocephaly and developmental delay are features of Sotos syndrome, an autosomal dominant disorder caused by variants in the gene NSD1. There is an increased risk for a variety of cancers including sacrococcygeal teratoma, neuroblastoma, acute lymphoblastic leukemia, small-cell lung cancer, and astrocytoma. Patients may also have a distinctive facial appearance (broad forehead, sparse frontotemporal hair, downslanting palpebral fissures, long and narrow face, long chin), and tall stature. Macrocephaly and developmental delay are also seen in association with PTEN hamartoma tumor syndrome (PHTS), which increases the risk for thyroid, breast, kidney, endometrial, and colon cancer.
💡For Sotos syndrome, remember "SOTT♡SSSSS": 'Serebral' gigantism, Overgrowth, Tall stature, Cardiac defects (♡), Scoliosis, Seizures, SET domain protein NSD1, 5q35 (looks like 'Sq3S').
Learning objective
Pathogenic variants in the BRCA1 and BRCA2 genes cause an autosomal dominant hereditary cancer predisposition syndrome that increases the risk for adult-onset breast, ovarian, pancreatic, and prostate cancer. Couples who are both carriers for BRCA2 are at risk for having a child with Fanconi syndrome, an autosomal recessive disorder that presents with forearm and thumb anomalies, growth failure, cytopenias, and childhood-onset cancers. BRCA1 & BRCA2 encode proteins that are involved in double-stranded DNA break repair, a mechanism that prevents the accumulation of mutations that lead to cancer.
2023 ABMGG General Exam Blueprint | VIII. Cancer genetics → b. Hereditary cancer predisposition → i. Autosomal dominant cancer predisposition syndromes & ii. Autosomal recessive cancer predisposition syndromes (page 4)



