Hello,
This week’s questions highlight a disorder with intermittent gross motor symptoms. This is the 3rd of 3 posts in a series on neurogenetics. I also uploaded a YouTube video related to the topic of this newsletter post.
Please feel free to reach out with any feedback or ideas for future posts (daniel@studyrare.com).
If you find this content valuable and would like to support my work, consider buying me a coffee. You can also forward this newsletter to genetic counseling students, clinical genetics fellows, or anyone interested in learning more about genetics and rare diseases.
Have a great week!
-Daniel
Questions
Question 63
A 11-month-old boy presents with intermittent episodes of paralysis. In the most recent episode, his right arm and leg were affected, but in prior episodes, it was his left arm and leg. These episodes typically last for 6 to 8 hours, and there is complete recovery back to his baseline between episodes. Developmentally, he rolls from front to back and does not yet pull to stand. A brain MRI performed in between episodes is normal. An EEG showed contralateral slowing during an episode without signs of seizures. There is no family history of similar symptoms. What is the most likely diagnosis?
Question 64
The disorder mentioned in Question 63 is inherited in which of the following manners?
Explanations
Question 63: Alternating hemiplegia of childhood
Question 64: Autosomal dominant
The patient in question 63 has alternating hemiplegia of childhood (AHC), a disorder that typically presents in children <18 months of age with recurrent episodes of hemiplegia (where one side of the body experiences weakness or paralysis) and developmental delays (manifesting in this patient as gross motor delays). These episodes can last from minutes to hours and may involve different sides of the body during different episodes (hence the word “alternating” in AHC), as seen in this patient. Episodes can be triggered by stressors such as poor sleep, fasting, or illness. The hemiplegia typically improves with sleep, and patients recover their baseline motor function between episodes. As was the case in this patient, a brain MRI is often normal, and EEG does not show epileptiform activity during the episodes (suggesting that the hemiplegia is not the result of a seizure). Infants and children with AHC typically have some degree of developmental delay, and up to half of children with AHC will eventually develop epilepsy.
Regarding management of AHC, flunarizine (a calcium channel blocker) can help reduce the severity and frequency of episodes in some patients. Patients should avoid triggers and maintain a regular sleep schedule. An ECG to rule out cardiac conduction anomalies should also be considered, as 60% of patients with AHC have an abnormal ECG.
💡The fact that AHC is associated with both neurologic and cardiac findings is consistent with the observation that ATP1A3 is expressed mainly in two tissues — the brain🧠 and the heart🫀.
Mechanism of disease
AHC is caused by pathogenic missense variants in ATP1A3, which encodes the sodium-potassium ATPase (Na/K-ATPase) pump. This Na/K-ATPase helps maintain the balance of sodium and potassium ions in neurons and cardiac cells. Three recurrent missense variants that act in a dominant negative manner account for ~60% of all AHC cases. Variants in ATP1A3 can disrupt ion transport, resulting in the neurological symptoms observed in AHC.
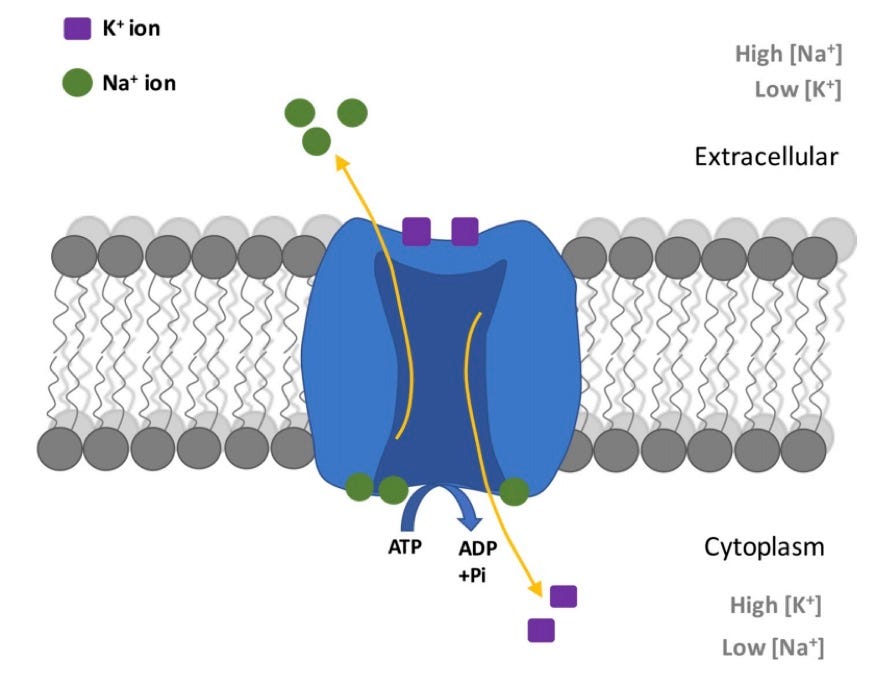
Missense variants in ion channels and dominant negative effects
Many ion channels are sensitive to missense variants that can result in a variety of neurological and/or cardiac disorders (as these organs are highly dependent on electrical impulses that are maintained by ion gradients). A de novo missense variant at a conserved amino acid in an ion channel should raise concern about the possibility of pathogenicity via a loss-of-function (if the encoded protein is a monomer, as with CFTR) or a dominant negative effect (if the encoded protein is a multimer, as with the Na/K-ATPase). This is especially true if this variant is absent in the general population and the patient has symptoms consistent with the expected phenotype. Dominant negative effects occur when a heterozygous variant encoding an abnormal protein (often non- or sub-functional) interferes with the function of the normal protein expressed from the other allele. This interference can results from the mutant protein forming nonfunctional complexes or disrupting cellular processes, leading to an abnormal phenotype, even when one copy of the normal allele is present.
ATP1A3-related neurologic disorders
AHC is part of a spectrum of ATP1A3-related neurologic disorders. The two other ATP1A3-related disorders include rapid-onset dystonia-parkinsonism (RDP) and cerebellar ataxia, areflexia, pes cavus, optic atrophy, and sensorineural hearing loss (CAPOS). As the name suggests, RDP is characterized by acute-onset dystonia, dysarthria, dysphagia, and bradykinesia (a feature of Parkinsonism) in children or adults. Motor symptoms can be also triggered by stressors (e.g. infection, alcohol) and do not typically respond to levodopa (vs sporadic Parkinson’s disease, where movement symptoms do typically respond to levodopa). CAPOS usually presents in infancy or early childhood with episodic ataxia and/or weakness triggered by a febrile illness.
Incorrect answers (Question 63)
Hypokalemic periodic paralysis (HPP) presents with intermittent paralysis and low potassium levels. However, HPP typically does not present in infants, is not usually associated with developmental delays, and usually affects both sides of the body simultaneously. In contrast, AHC exhibits alternating hemiplegia (only one side of the body at a time).
Spastic paraplegia 4 (SPAST-HSP) is a form of hereditary spastic paraplegia characterized by progressive stiffness and weakness in the legs that most often manifests in young adults. Patients with SPAST-HSP have neurologic symptoms that are constant and progressive rather than episodic, as seen in AHC.
Familial hemiplegic migraine is characterized by migraines preceded by a hemiplegic aura. Patients may also have other accompanying symptoms that precede the migraine including changes in vision, difficulty with speech, and numbness or paresthesias of the extremities. Most cases of hemiplegic migraine do not have an identifiable underlying genetic etiology, though a subset of patients have variants in ATP1A2, CACNA1A, or SCN1A. Though some patients with CACNA1A-related hemiplegic migraine have developmental delays, it would be more typical for the hemiplegia in these cases to present after infancy (e.g. later in the 1st or 2nd decade of life).
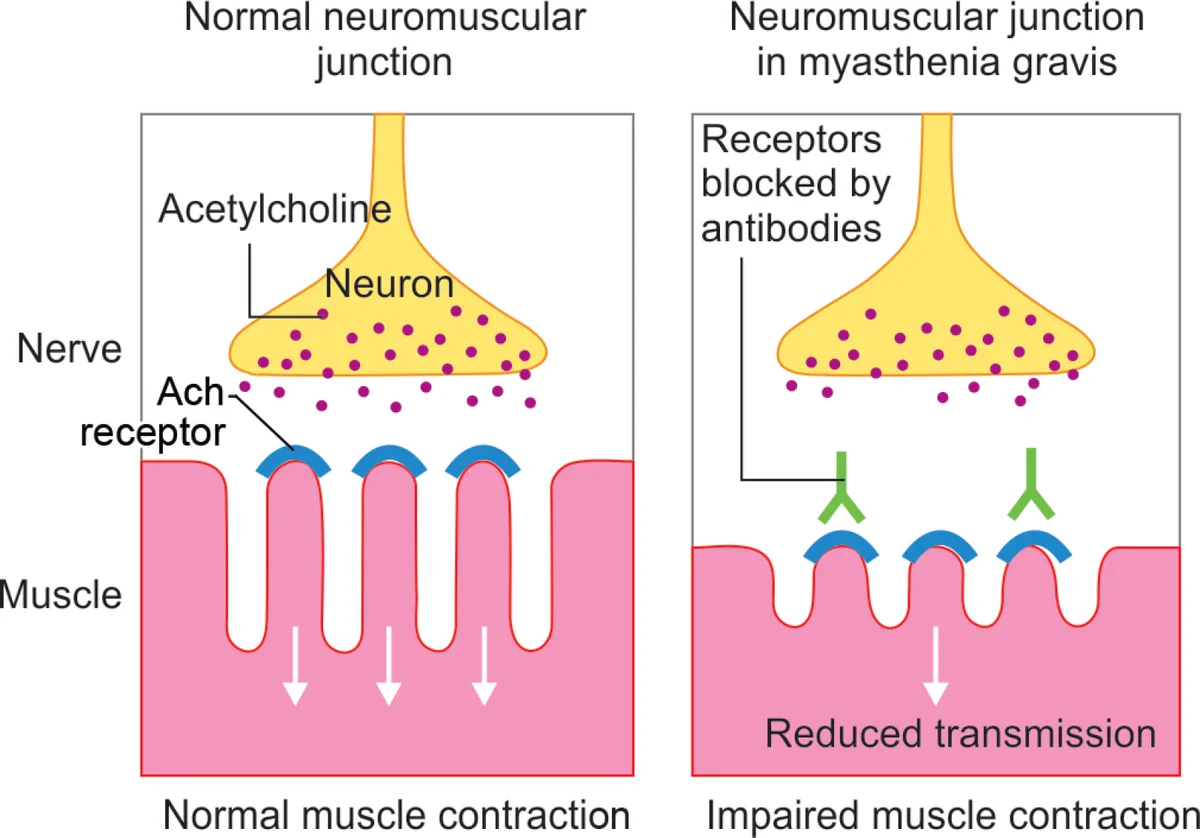
CHRNE-related congenital myasthenia typically presents with muscle weakness and fatiguability that worsens with exercise rather than the characteristic alternating hemiplegia seen in AHC. CHRNE encodes the epsilon subunit of the acetylcholine receptor (AChR), which is important for neurotransmission at the neuromuscular junction (NMJ). Antibodies that bind to the AChR are the cause of myasthenia gravis, an autoimmune disorder that presents in adults with fatiguability that worsens with exercise. Patients with both CHRNE-related congenital myasthenia and myasthenia gravis can be treated with an acetylcholinesterase inhibitor that prevents acetylcholine breakdown, thereby increasing the amount of acetylcholine at the NMJ.
Learning objective
Alternating hemiplegia of childhood (AHC) presents in infants with recurrent episodes of acute-onset unilateral and transient weakness that can be triggered by stressors (e.g. illness, fasting). Most cases of AHC are caused by missense, dominant-negative variants in ATP1A3, which encodes the sodium-potassium ATPase. AHC is inherited in an autosomal dominant manner. Many patients with AHC have developmental delays, more than half have ECG abnormalities, and nearly half of patients with AHC will develop seizures.
2023 ABMGG General Exam Blueprint | V. Single Gene Inheritance → d. Single Gene Disorders → v. Neurogenetic disorders
2023 ABGC Exam Content Outline | Domain 1. Clinical Information, Human Development, and Genetic Conditions → C. Genetic Conditions → 1. Clinical features & 7. Mode of inheritance